Mass spectroscopy and Ar calculations ![]()
1) A
sample of sulphur has three isotopes: sulphur-32, sulphur-33 and sulphur-34.
Calculate the relative atomic mass from the data below: Give your answer to the appropriate number of
decimals places.
|
percentage abundance |
95.02 |
0.76 |
4.22 |
|
Isotopic mass |
32 |
33 |
34 |
RAM = S (Abundance x m/z)
Total Abundance
RAM = (95.02 x 32) +
(0.76 x 33) + (4.22 x 34)
(95.02 +
0.76 + 4.22)
RAM = 32.09
(2dp as question is to 2dp)
2) Boron has 2 isotopes and the
mass spectrum is shown below. Calculate
its relative atomic mass:
|
|
RAM = (1
x 10) + (4 x 11) (1 + 4) RAM =
10.8 |
3) Lithium has two naturally occurring isotopes 6Li
and 7Li. Calculate the percentage abundance of each isotopes given
that the Ar of Lithium is 6.9. Show your working.
6.9 is 9/10
th's between 6Li
and 7Li
So 90 % must be 7Li so 10
% must be 6Li
OR:
RAM = S (Abundance x m/z)
Total Abundance
6.9 = (A x 6) +
((100-A) x 7)) Where
A is the Abundance of 6Li
(A +
100 - A)
6.9 = 6A +
(7(100-A))
100
100 x 6.9 = 6A +
700 - 7A
690 = 6A +
700 - 7A
690 -700 = 6A
- 7A
-10 = -A
A = 10 %
4) A specific alloy of bronze has been developed
for use in aircraft due to its high strength and resistance to corrosion.
It is an alloy of two metals and ts mass spectrum is shown below. Identify the two metals in
this bronze and explain why there are three peaks.
|
|
Al, 27Al Cu with 2 isotopes, 63Cu and 65Cu |
5) For the above question,
calculate the relative atomic mass of the element with Isotopes. For copper ONLY
RAM = S (Abundance x m/z)
Total Abundance
RAM = (124 x 63) + (56
x 65)
(124 + 56)
RAM = 63.6
6) A
sample of magnesium has three isotopes: magnesium-24, magnesium -25 and
magnesium -26. Given that the relative atomic mass of magnesium is 24.3, calculate
the relative abundance for magnesium 24 from the data below:
|
Relative abundance |
|
100 |
110 |
|
Isotopic mass |
24 |
25 |
26 |
RAM = (A x 24) + (100
x 25) +
(110 x 26) Where A
is the abundance of 24Mg
(A +
100 + 110)
24.3 = 24A + 2500 + 2860
(A +
210)
24.3(A + 210)
= 24A + 5360
24.3A + 5103 =
24A + 5360
24.3A - 24A =
5360 - 5103
0.3A = 257
A = 257
0.3
A = 857
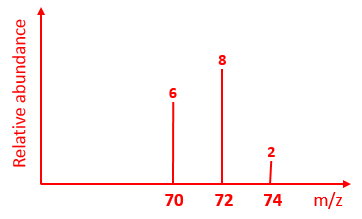
7) Sketch the mass spectrum you
would likely see for Chlorine molecules
given the following data:
|
Relative abundance |
3 |
1 |
|
Isotopic mass |
35 |
37 |
|
|
Possible combinations of the molecules Cl2+
: 35Cl – 35Cl+ Total mass = 70, total abundance = 6 (3+3) 37Cl – 35Cl+ and 35Cl – 37Cl+ (ie 2
scenarios likely) Total mass = 72, Total abundance = 8 (1+3) + (3+1) 37Cl – 37Cl+ Total mass = 74, total abundance = 2 (1+1) |
8) Mass spectroscopy is a
technique used to determine relative abundancies of isotopes of elements.
a) Briefly explain the stages in mass spectroscopy:
|
Ionisation |
Atoms / molecules are ionised by losing an electron. |
|
Acceleration |
The ions are accelerated using a negatively charged plate. They are all given the same kinetic
energy. The speed at which the ion
will travel will depend upon the mass of the ion, m/z |
|
Ion drift |
Time of flight: Higher mass
ions (m/z) move slower than lower mass ions (m/z) |
|
Detector |
The ions with a smaller mass reach the detector first. The ion picks up an electron. |
b) Write an equation showing the ionisation
stage, use M to represent an atom:
M à M+ +
e-
c) Write an equation to show what happens at the
detector, use M to represent an atom:
M+ + e-
à M