1B Fats and oils /30
|
1B Fats and oils /30 |
|
1) This question is about fatty acids and fats:
a) Draw and name the fatty acid 18:2(9.12) [2]
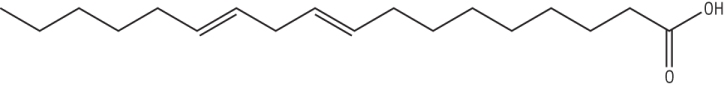
octadec 9,12 dienoic acid
b) Use the shorthand notation for the following molecule [1]
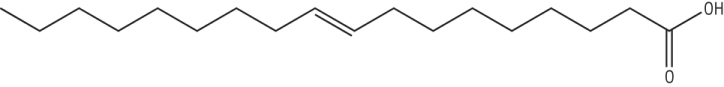
18:1(9)
c) Name the functional groups in the molecule in (b) [2]
carboxylic acid [1] alkene [1]
d) Describe a simple chemical test you could carry out to distinguish between the molecules below [2]
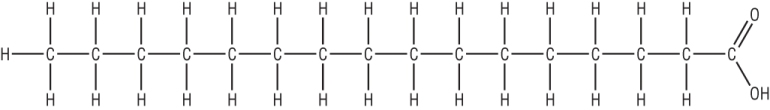

Bromine water [1] 2nd molecule will change it from orange à clear and colourless [1]
e) Write a balanced chemical equation for the reaction between glycerol and hexadecanoic acid.
Circle the new functional group and label it [3]
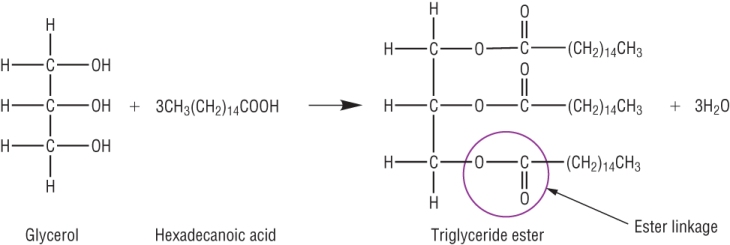
Triester formula correct [1] Rest of equation correct [1] ester group circled and named [1]
2) This question is about fats and oils:
a) Draw the skeletal formulas for a i) saturated fatty acid ii) trans fatty acid iii) cis fatty acid [3]
i) skeletal formula with no double bonds but a carboxylic acid functional group at the end [1]
ii) skeletal formula with at least 1 double bond, molecule still flat (ish), carboxylic acid at end [1]
iii) skeletal formula with at least 1 double bond, molecule in a 'v' shape, carboxylic acid at end [1]
b) Use your answers in (a) to explain the health issues associated with each (i) - (iii) [8]
i) and ii) efficient packing [1] strong VDW [1] solid causes blockages [1] heart disease [1]
iii)inefficient packing [1] weak VDW [1] liquid [1] blockage and heart disease avoided [1]
c) Explain the difference between and fat and an oil [3]
Fats are solids and oils are liquids [1] fats have stronger IMF of attraction [1]
bringing the melting point above room temperature [1] ORA
d) Use your knowledge of the alkenes to describe how solids (margarine) are formed from oils [3]
C=C are removed by reaction with H2 [1] with a nickel catalyst [1] 15oC [1]
3) Oils can be used to produce fuels.
a) Draw the structure of an oil made between glycerol and hexadecanoic acid: [1]
Hex%20acid.jpg)
b) Write an equation for the formation of biodiesel from the triester in (a) [2]
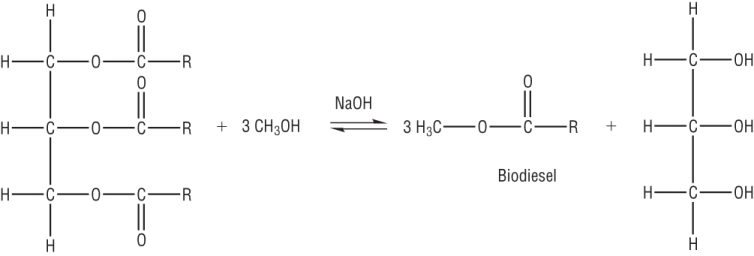
Methyl hexadecanoate [1]
Rest of the equation [1]