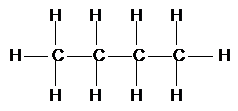
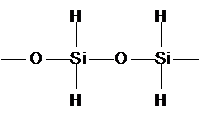
Introduction to organic chemistry
Organic Chemistry is the study of carbon chemistry as carbon has an amazing ability to join together in chains, rings, balls etc.
Pre – petrochemical industry almost all carbon compounds were extracted from living things. It was Lavoisier in 1784 who first suggested that all compounds extracted from living things contain carbon and hydrogen and are called Hydrocarbons.
Carbon also joins with other elements easily such as oxygen, hydrogen, nitrogen, phosphorous and the halogens.
Hydrocarbons make up over 90% of all known compounds.
Carbon can join in many different ways and shapes.
Organic compounds also contain other elements giving rise to functional groups (later).
Why carbon and not silicon?
|
|
|
 |
 |
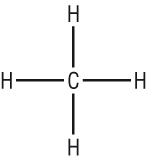
| 4 bonds only | A double bond and 2 single bonds to hydrogen = 4 |
 |
 |
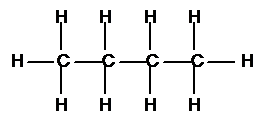
| Saturated - is a hydrocarbon with single bonds only | Unsaturated - is a hydrocarbon with carbon carbon multiple bonds |
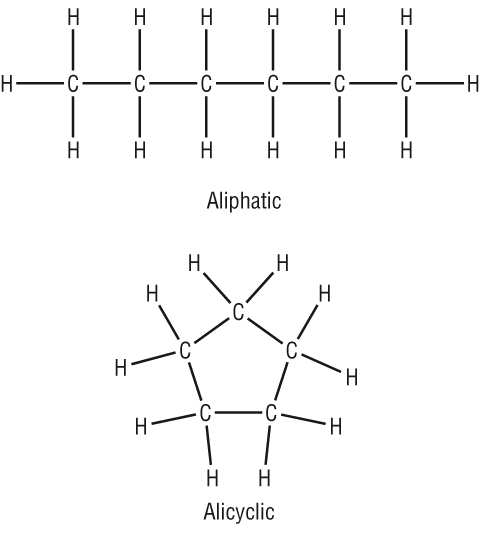 |
Aliphatic - is a hydrocarbon in which the carbon atoms are all joined in a straight lines (branched or unbranched) |
| Alicyclic - is a hydrocarbon in which the carbon atoms are joined together in a ring structure |
|
No of C's |
Name |
Formula |
|
1 |
Methane |
CH4 |
| 2 |
Ethane |
C2H6 |
| 3 |
Propane |
C3H8 |
| 4 |
Butane |
C4H10 |
| 5 | Pentane |
C5H12 |
| 6 | Hexane |
C6H14 |
| 7 | Heptane |
C7H16 |
| 8 | Octane |
C8H18 |
| 9 | Nonane |
C9H20 |
| 10 | Decane |
C10H22 |
Because there are so many organic compounds we have to have a systematic way of naming them.
This is called Nomenclature
Organic molecules are usually made up from:
Carbon chain
Side chains (alkyl groups)
Functional groups
Stems, prefix and suffix:
Stem The longest carbon chain - the main name (in the middle) (carbon chain)
Prefix Added before the main name - pre - main name (side chains)
Suffix Added after the main name - post - main name (functional groups)
Alkyl groups:
If you remove a hydrogen from an alkane you have a group that has a bond that can join to the main carbon chain.
Based on the alkanes the ending of these are changed to alkyl
 |
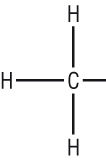 |
| Methane | Methyl |
The first six alkyl side chains are in the table below:
|
No of C's |
Name |
Formula |
|
1 |
Methyl |
CH3 - |
| 2 |
Ethyl |
C2H5 - |
| 3 |
Propyl |
C3H7 - |
| 4 |
Butyl |
C4H9 - |
| 5 | Pentyl |
C5H11 - |
| 6 | Hexyl |
C6H13 - |
Naming alkanes:-
1) Look for the longest carbon chain alkane
2) Look for the functional groups
3) Look for the position of the functional group and assign a number. Use the lowest number possible counting from one end of the carbon chain.
4) The name goes in reverse order of the 3 points above.
Example:-
|
|
1) Look for the longest straight chain alkane
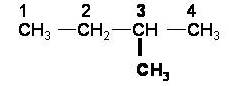 |
The longest carbon chain is in bold. It is 4 carbons long which makes it butane |
| ____________Butane |
2) Look for the functional groups
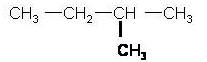 |
A functional group is any side chain off the straight chain. CH3 is a derivative of methane (CH4) which we call methyl. See the table below |
| _______Methylbutane |
3) Look for the position of the functional group and assign a number. Use the lowest number possible counting from one end of the carbon chain.
 |
3 – Methylbutane. This is incorrect because if we number from the other end we get a lower number. |
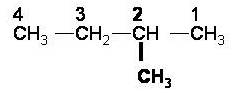 |
Numbering from the other end we get 2 – Methylbutane. This is correct because if we get a lower number. |
| 2 – Methylbutane |
4) The name goes in reverse order of the 3 points above.
2 - Methyl butane
(3) (2) (1)
5) Additional side chains or stems
If there is more than 1 side chain we write the names in alphabetical order:-
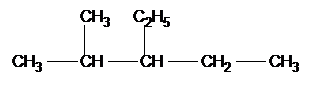 |
3-ethyl 2 methyl pentane |
If there is more than 1 chain on the same carbon we use di, tri, tetra etc:
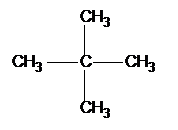 |
2,2-dimethyl propane |
Naming alkenes
These contain a C=C, the ending of the name changes to ‘ene’ and we have to put a number to where the double bond is in the carbon chain:-
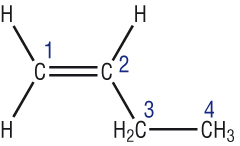 |
But – 1 – ene |
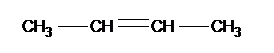 |
But – 2 – ene |
Questions 1-2 P105
Naming compounds with functional groups
Organic Chemistry is studied in a systematic way because each different group of atoms attached to a carbon atom has its own characteristic set of reactions.
These are known as Functional groups:-
|
Functional group |
Formula |
Prefix (side chains) |
Suffix (functional group) |
|
Alkane |
C - C
|
-ane |
|
|
Alkene |
C = C
|
-ene |
|
|
Halogenoalkane |
– F |
Floro - |
|
|
– Cl |
Chloro - |
||
|
– Br |
Bromo - |
||
|
– I |
Iodo - |
||
|
Alcohols |
– OH
|
Hydroxy - (if other functional groups are present)
|
- ol |
|
Aldehydes |
|
- al |
|
|
Ketones |
|
- one |
|
|
Carboxyllic acids |
|
- oic acid |
If a suffix starts with a vowel then the stem has 'an' added
Names for alkanes containing a ring of carbon atoms
If an alkane is cyclic we use the prefix ‘Cyclo’

cyclohexane
Names for Halogenoalkanes
|
|
Longest chain = 3C = prop Functional group = Cl (prefix) = Chloroprop Chloro is on carbon 1 = 1 chloroprop No suffix = ane = 1 chloropropane |
Names for alcohols
|
|
Longest chain = 5C = pent Functional group =OH (suffix starts with a vowel) = pentan ol OH is on carbon 3 = pentan - 3 - ol |
Names for aldehydes
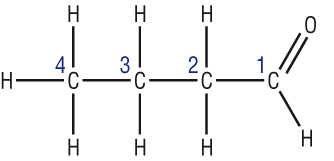 |
Longest chain = 4C = but Functional group = CHO (suffix starts with a vowel) = butanal The 'al' does not need a number as all aldehydes are at the end of the molecule. |
More than one of the same type of functional group
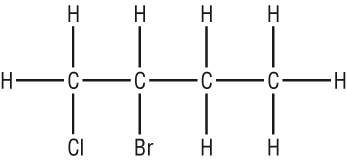 |
Longest chain = 4C = but Functional group = Cl on carbon 1 (prefix) = 1 - chloro Functional group = Br on carbon 2 (prefix)n = 2 - bromo Functional groups are named alphabetically: 2 - bromo - 1 - chlorobut No suffix = ane = 2 - bromo - 1 - chlorobutane |
Names for 2 different functional groups:
 |
Longest chain = 3C = prop Functional group = OH on carbon 1 (suffix, starts with a vowel so add 'an') = propan - 1 - ol Side chain = CH3 on carbon 2 (prefix) = 2 - methylpropan - 1 - ol |
Names for many of the same functional groups:
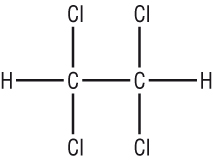 |
Longest chain = 2C = eth Functional group = Cl, (2 x on carbon 1) and 2 x on carbon 2 (prefix), numbers first then how many chlorines: 1,1,2,2 - tetrachloroeth No suffix = ane = 1,1,2,2 - tetrachloroethane |
Note: di = 2, tri = 3, tetra = 4
Question 1 P 107
Formulae of organic compounds:
Empirical formula:
Definition: Empirical Formula is the simplest ratio of atoms of elements in a compound.
Molecular formulae
Definition: Molecular formulae is the actual ratio of atoms of elements in a compound.
This can be calculated using moles from percentage composition:-
Recap from Module 1:
Example 1: A sample of iron oxide was found to have 11.2g of iron and 4.8g of oxygen. Calculate the formula of this compound:
Fe O
11.2 4.8
Divide by Ar 11.2/56 4.8/16
Moles 0.2 0.3
Divide by smallest 0.2/0.2 0.3/0.2
Ratio 1 : 1.5
Ratio 2 : 3
Fe2O3
Example 2
A sample of hydrocarbon was found to have 1.20g of carbon and 0.25g of hydrogen. Calculate the Empirical formula of this compound. Then find out the molecular formula if the Mr = 58
C H
1.20 0.25
Divide by Ar 1.2/12 0.25/1
Moles 0.1 0.25
Divide by smallest 0.1/0.1 0.25/0.1
Ratio 1 : 2.5
Ratio 2 : 5
Empirical formula C2H5 Mr = 29
Molecular formula C4H10 58 / 29 = 2, so double up the numbers of atoms to make the actual ratio
General Formula:
Definition: This is the simplest algebraic formula for a member of a homologous series (of the same functional group)
| Alkanes | CnH2n+2 | Alkenes | CnH2n | Alcohols | CnH2n+1OH | ||
| Methane | CH4 | Methanol | CH3OH | ||||
| Ethane | C2H6 | Ethene | C2H4 | Ethanol | C2H5OH | ||
| Propane | C3H8 | Propene | C3H6 | Propanol | C3H7OH |
Displayed formula:
Definition: Shows the relative positioning of all the atoms in a molecule, and all the bonds between them
 |
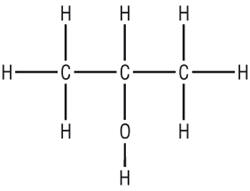 |
| Butane: All the atoms and bonds are shown | Propan-2-ol: Even O - H bonds are shown |
Questions 1-3 P109
Structural and skeletal formula:
Structural formula:
Definition: shows the minimum detail for the arrangement of atoms in a molecule
Typically we show the ratio of all the atoms attached to each carbon atom in a molecule:
 |
 |
| Butane:
CH3CH2CH2CH3 |
1 - Chloropropane:
CH3CH2CH2Cl |
With many CH2's we can put them in brackets:
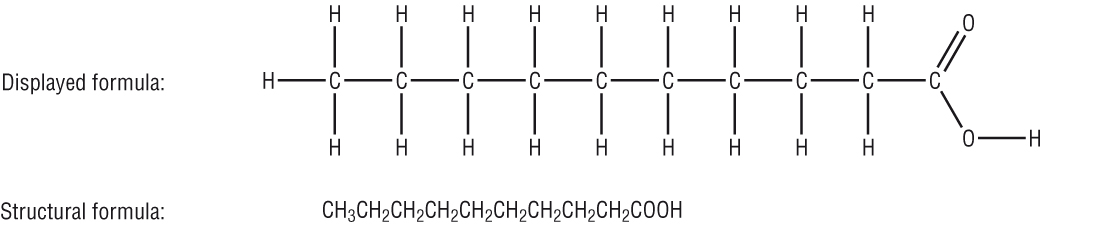 |
|
This can be simplified to: CH3(CH2)8COOH |
Skeletal formula:
Definition: The hydrogen's are removed leaving a carbon skeleton and associated functional groups
A good way to approach this is to count and number the carbons. This can then be transposed to the carbon skeleton:
|
|
Other examples:
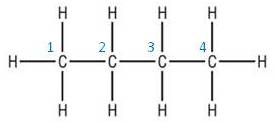 |
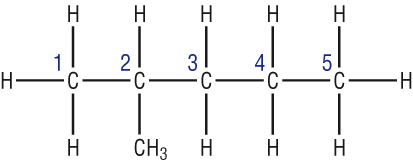 |
| Structural formula:CH3CH2CH2CH3 | Structural formula:CH3CH(CH3)CH2CH2CH3 |
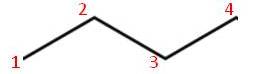 |
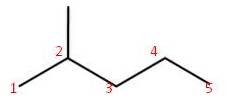 |
No carbon or hydrogen atoms are shown
A carbon atom exists at the ends
A carbon atom exists at each point
Cyclic compounds:
Cyclic compounds are usually drawn as skeletal:
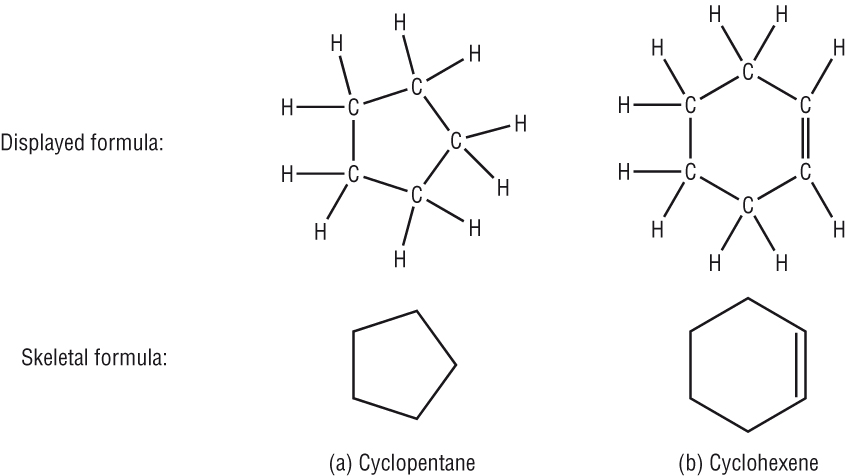 |
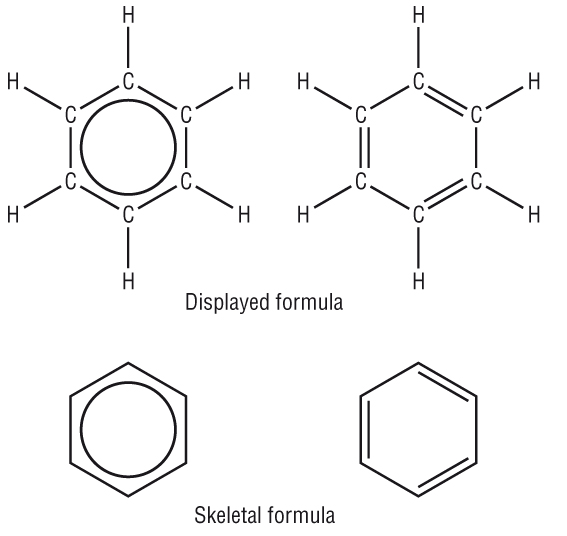 |
Questions: 1-3 P111
Skeletal formulae and functional groups
Unsaturated hydrocarbons:
Add numbers to the carbons on the skeletal formulae. This is how many carbons in the molecule.
Add a double bond between the corresponding carbons from the skeletal to the displayed:
| Skeletal formulae: | Fill in the displayed formula: |
|
|
|
| Pent-2-ene |
Compounds with functional groups:
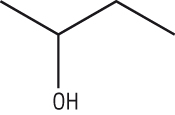
Butan-2-ol:
Functional groups must be included in skeletal formulae
The end of the side chain off carbon 2 is now an OH instead of a CH3
| Skeletal formulae: | Fill in the displayed formula: |
 |
|
| Butan-2-ol |
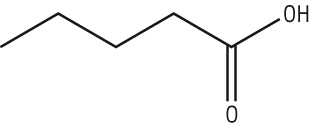
Pentanoic acid:
Coming off the final carbon is an =O and an -OH, these must be added to the end carbon:
| Skeletal formulae: | Fill in the displayed formula: |
 |
|
| pentanoic acid |
More complex AS / A2 structures:
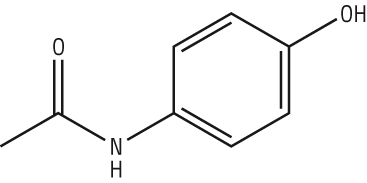 |
Paracetamol |
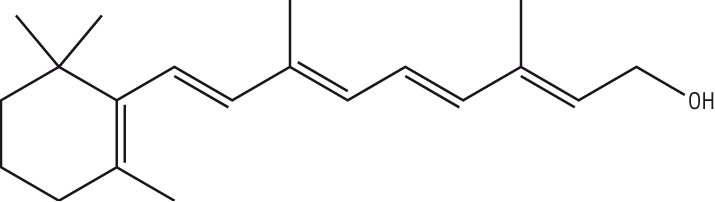 |
Retinol, vitamin A |
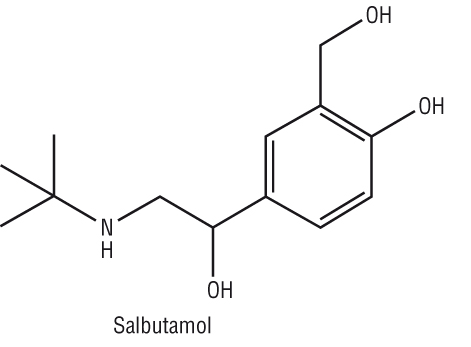 |
Salbutamol |
Note:
If a carbon in the skeletal structure is replaced with another element then we have to write the symbol of that element in its place
Questions: 1-4 P113
Structural isomers:
Activity 1:
Use the molymods to make and draw as many molecules as possible using all of 5 carbons and 12 hydrogens, C5H12.
There are 3 different shapes.
All of the molecules above contain the same number of atoms but they are arranged differently. These are called structural isomers.
This is one reason why we use a systematic method to name organic molecules.
Definition: These are compounds with the same molecular formula but with different structural arrangements of atoms
Activity 2:
Using the molymods make and draw as many molecules as possible using 3 carbons, 8 hydrogens, and 1 oxygen, C3H8O.
Some of these structures that you have made will be from different homologous groups / have different functional groups.
This means that although they have the same empirical formula, they will have very different physical and chemical properties.
Activity 3:
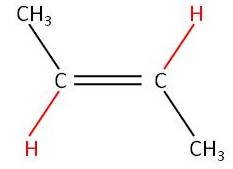 |
No free rotation around the C=C |
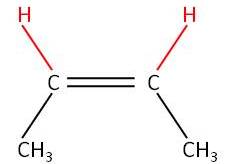 |
| Trans
but-2-ene (E-but-2-ene) |
Cis
but-2-ene (Z-but-2-ene) |
|
|
1) Homolytic fission
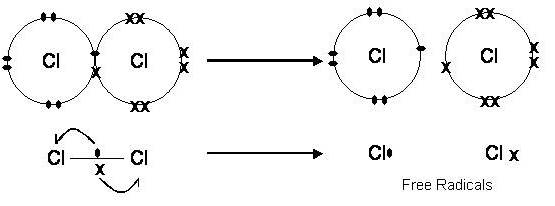
Heterolytic fission
|
|
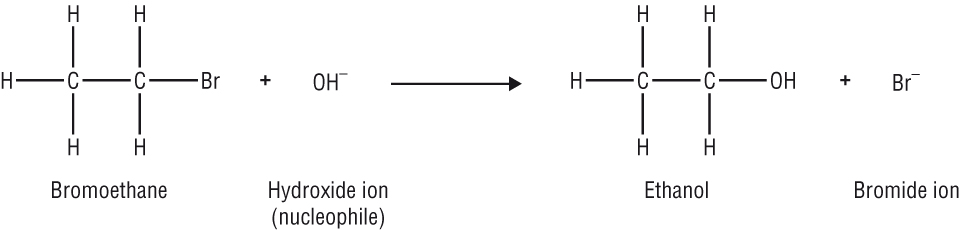
2) Nucleophiles: these are attracted to electron deficient atom, d+ and donate a pair of electrons to form a new covalent bond
These are often negative ions but must have a lone pair of electrons as these are donated to form a new covalent bond.
Br-, OH-, H2O, NH3
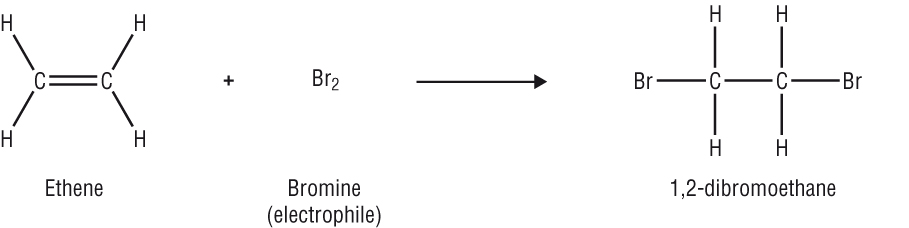
3) Electrophiles: these are attracted to electron rich atom, d- and accept a pair of electrons to form a new covalent bond
These are often positive ions.
Br2, HBr, NO2+
 |
 |
 |