1) Define the term first ionisation energy. [3]
2) Electrons are arranged in energy levels.
Complete
the table below to show how many electrons completely fill each of the
following. [3]
|
|
number of
electrons |
|
a d
orbital |
|
|
a p
sub-shell |
|
|
the third
shell (n = 3) |
|
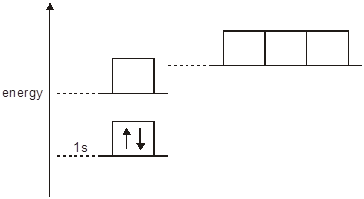
3) The energy diagram below is for
the eight electrons in an oxygen atom. The diagram is incomplete as it only
shows the two electrons in the 1s level.
Complete the diagram for the oxygen atom by:
(a) adding labels for
the other sub-shell levels, [1]
(b) adding arrows to show how
the other electrons are arranged. [1]
4) The table shows the first 6 successive ionisation
energies of an element X, which is in Period 3 of the Periodic
Table.
Use the table to identify element
X. Explain how you decided on your answer. [3]
|
element |
ionisation
energy / kJ mol–1 |
|||||
|
|
1st |
2nd |
3rd |
4th |
5th |
6th |
|
X |
578 |
1817 |
2745 |
11 578 |
14 831 |
18 378 |
5) (a)
Write an equation, with state symbols, to represent the second ionisation
energy of calcium. [2]
(b) Why is the second ionisation energy of
calcium greater than their first ionisation energies? [1]
(c) Explain why the first ionisation energy of
strontium are less than that of calcium. [3]
6) The electronic configuration of
a bromine atom can be written in terms of sub-shells.
(a) Complete the electronic configuration of a
bromine atom. [2]
1s22s22p63s23p6 ...............
(b) Why is bromine classified as a p-block
element? [1]
(c) Write
the electron configuration for a bromine ion [1]
1s22s22p63s23p6 ...............