Oxidation involves the movement of electrons
This can be monitored using oxidation numbers from AS:
| 1) | Element | = 0 |
| 2) | Oxide | = -2 |
| 3) | Hydrogen | = +1 |
| 4) | Ionic element | = charge on ion |
Redox reactions:
These are reactions where both oxidation and reduction occur
Remember:
| Oxidation: | Reduction | |
| Loss of e | Gain of e | |
| Ox No increases | Ox No decreases | |
| Are reducing AGENTS | Are oxidizing AGENTS |
Constructing redox equations using relevant half - equations
This is done by balancing the numbers of electrons lost by one half equation with those being gained by another half equation
Example:
Iron reacts with copper (II) ions forming irone (III) and copper:
Step 1: Identify the redox half equations:
| Fe | à | Fe3+ | + | 3e- | |||||
| Cu2+ | + | 2e- | à | Cu |
Step 2: Balance the electrons:
| 2Fe | à | 2Fe3+ | + | 6e- | x2 | |||
| 3Cu2+ | + | 6e- | à | 3Cu | x3 |
Step 3: Add the half equations together and cancel out the electrons:
| 2Fe | à | 2Fe3+ | + | 6e- | ||||||||
| 3Cu2+ | + | 6e- | à | 3Cu | ||||||||
| 2Fe | + | 3Cu2+ | + |
|
à | 3Cu | + | 2Fe3+ | + |
|
||
This gives:
| 2Fe | + | 3Cu2+ | à | 3Cu | + | 2Fe3+ |
Constructing redox equations using oxidation numbers:
Example:
Hydrogen iodide, HI is oxidised to iodine, I2 by concentrated sulphuric acid, H2SO4, which is reduced to hydrogen sulphide, H2S
Step 1: Identify the redox half equations:
| HI | à | 1/2 I2 | + | e- | |||||
| H2SO4 | + | 8e- | à | H2S |
Step 2: Balance the electrons:
| 8HI | à | 4I2 | + | 8e- | x8 | ||||
| H2SO4 | + | 8e- | à | H2S |
Step 3: Add the half equations together and cancel out the electrons:
| 8HI | à | 4I2 | + | 8e- | ||||||||
| H2SO4 | + | 8e- | à | H2S | ||||||||
| 8HI | + | H2SO4 | + |
|
à | 4I2 | + | H2S | + |
|
||
Step 4: Balance the oxygen's by adding water to the opposite side:
| 8HI | + | H2SO4 | à | 4I2 | + | H2S | + | 4H2O | |
| O | 4 | 0 | so | add 4 x waters |
Step 5: Balance the hydrogen's by adding hydrogen ions to the opposite side:
| 8HI | + | H2SO4 | à | 4I2 | + | H2S | + | 4H2O | |
| H | 10 | 10 |
Hints for completing redox reactions:
| In acidic conditions | Balance using H+ | |
| In alkaline conditions | Balance using OH- |
Summary:
Step 1: Identify the redox half equations:
Step 2: Balance the electrons:
Step 3: Add the half equations together and cancel out the electrons:
Step 4: Balance the oxygen's by adding water to the opposite side:
Step 5: Balance the hydrogen's by adding hydrogen ions to the opposite side:
| In acidic conditions | Balance using H+ | |
| In alkaline conditions | Balance using OH- |
Qu 1-2 P 183
The reactivity series (from GCSE)
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
Consider the following reactions:
A) Zinc powder is added to a solution of copper sulphate:
As zinc is more reactive than copper, the reaction proceeds and Zn donates its electrons to Cu2+
Zn(s) + Cu2+(aq) à Zn2+(aq) + Cu(s)
The half equations:
Zn(s) à Zn2+(aq) + 2e- Reaction 1
Cu2+(aq) + 2e- à Cu(s) Reaction 2
In reaction 1 the oxidation number of zinc increases from 0 à +2. Zinc is oxidised.
In reaction 2 the oxidation number of copper decreases from +2 à 0. Copper is reduced.
B) Magnesium powder is added to a solution of zinc sulphate:
As Magnesium is more reactive than zinc, the reaction proceeds and Mg donates its electrons to Zn2+
Mg(s) + Zn2+(aq) à Mg2+(s) + Zn(s)
The half equations:-
Mg(s) à Mg2+(aq) + 2e- Reaction 1
Zn2+(aq) + 2e- à Zn(s) Reaction 2
In reaction 1 the oxidation number of magnesium increases from 0 à +2. Magnesium is oxidised.
In reaction 2 the oxidation number of zinc decreases from +2 à 0. Zinc is reduced.
In these 2 examples (A and B) you have seen that zinc has been oxidised and reduced.
This means that the zinc reactions can be treated as an equilibrium:
| Zn(s) | Zn2+(aq) | + | 2e- |
Applying Le Chateliers Principle:-
1. Add electrons to the system and the equilibrium will shift so as to remove electrons.
2. Remove electrons from the system and the equilibrium will shift so as to produce electrons.
We call this is metal / metal ion system or half cell.
With a metal whose tendency to loose electrons is greater than that of zinc (Magnesium), the equilibrium will shift towards the reactants (Zn(s))
With a metal whose tendency to loose electrons is less than that of zinc (Copper), the equilibrium will shift towards the products (Zn2+(aq)).
The electrochemical series:
|
This means:-
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Will a reaction actually take place:
Example 1
1) Write out the half reactions. The more (-)ve value will produce electrons, the more positive will react with electrons.
| Mg2+(aq) | + | 2e- |
|
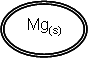 |
Eq = -2.37 |
1 |
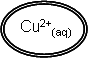 |
+ | 2e- |
|
Cu(s) | Eq = +0.34 |
2 |
From this rule you can easily cirlcle the reactants that you must have in order for the reaction to occurr.
It is possible to write a reaction from this because this tells you that reaction 1 is going to move to the LHS and reaction 2 is going to move to the RHS.
2) Draw the direction of each of the half reactions:
|
|
| Mg2+(aq) | + | 2e- |
|
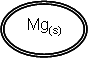 |
Eq = -2.37 |
1 |
||
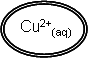 |
+ | 2e- |
|
Cu(s) | Eq = +0.34 |
2 |
|
|
3) Writing balanced redox equation
Write out the 2 half reactions in the direction they occur.
Mg(s) à Mg2+(aq) + 2e-
Cu2+(aq) + 2e- à Cu(s)
Balance the electrons (if necessary).
4) Add the half reactions
Add the reactions together and the electrons will cancel out:-
Mg(s) à Mg2+(aq) + 2e-
Cu2+(aq) + 2e- à Cu(s)
Mg(s) + Cu2+(aq) + 2e- à Mg2+(aq) + Cu(s) + 2e- electrons cancel out
Mg(s) + Cu2+(aq) à Mg2+(aq) + Cu(s)
This can now be extended to metal ion / metal ion systems and also non metal systems:
Example 2:
1) Write out the half reactions. The more (-)ve value will produce electrons, the more positive will react with electrons.
| Sn2+(aq) | + | 2e- |
|
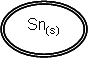 |
Eq = -0.14 |
1 |
||
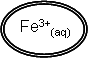 |
+ | e- |
|
Fe2+(aq) | Eq = +0.77 |
2 |
2) Draw the direction of each of the half reactions:
|
|
| Sn2+(aq) | + | 2e- |
|
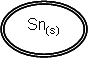 |
Eq = -0.14 |
1 |
||
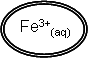 |
+ | e- |
|
Fe2+(aq) | Eq = +0.77 |
2 |
|
|
3) Writing balanced redox equation
Write out the 2 half reactions in the direction they occur.
Sn(s) à Sn2+(aq) + 2e-
Fe3+(aq) + e- à Fe2+(aq) x2
Balance the electrons.
4) Add the half reactions
Add the reactions together and the electrons will cancel out:-
Sn(s) à Sn2+(aq) + 2e-
2Fe3+(aq) + 2e- à 2Fe2+(aq)
Sn(s) + 2Fe3+(aq) + 2e- à Sn2+(aq) + 2Fe2+(aq) + 2e- electrons cancel out
Sn(s) + 2Fe3+(aq) à Sn2+(aq) + 2Fe2+(aq)
Summary steps:
1) Write out the half reactions. The more (-)ve value will produce electrons, the more positive will react with electrons.
2) Draw the direction of each of the half reactions:
3) Writing balanced redox equation
4) Add the half reactions
Limitations of predictions using standard electrode potentials:
a) Effect of changing concentration:
The symbol q represents standard conditions of 298k and 1 molar solutions.
Consider the half reaction:
Cu2+(aq) + 2e-
![]() Cu(s)
Cu(s)
Increasing the concentration of the Cu2+ ions moves the equilibrium to the RHS
This removes electrons from the equilibrium making it less negative
b) Effect of Eqcell value:
In practice the Eqcell values have to differ by at least 0.4v in order for the reaction to have enough of a difference in energy to overcome activation energies.
Qu 1 P189
Electricity from chemical reactions
We have seen so far that chemical reactions involve a transfer of electrons from one metal / metal ion system to another.
It is possible to make these electrons move from one system to another through an external wire - electrical current.
This is the basis of all batteries. The chemistry involved is called electrochemistry:
Cells and half cells:
Batteries are called cells and are based upon the redox reactions studied so far but with the electrons flowing through an external wire.
Zn(s) + Cu2+(aq) à Zn2+(aq) + Cu(s)
The redox reactions consist of 2 half reactions. We call these half cells (as 2 of them are needed for a cell)
The half equations:
Zn(s) à Zn2+(aq) + 2e- half cell: flows out of the system to the external wire
Cu2+(aq) + 2e- à Cu(s) half cell: flows into the system from the external wire
Consider the half cell:
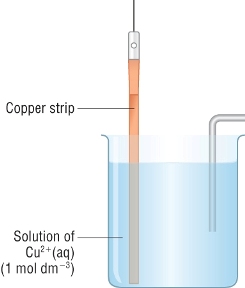 |
Cu2+(aq) + 2e- With a more negative half cell:
With a more positive half cell:
|
So what determines where the copper half cell fits in the electrochemical series?
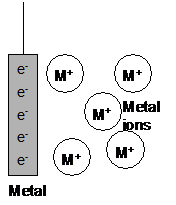
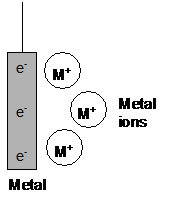
It is down to the tendency of a metal to form ions in solution.
When a metal strip is put into a solution of its ions, an equilibrium is set up:
| M(s) | Mz+(aq) | + | ze- | |
 |
|
|
|
 |
|
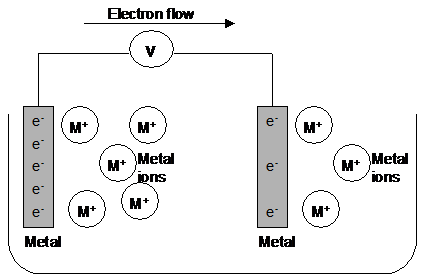
As electrons are removed from one metal/ion system, the equilibrium shifts to replace the electrons.
As electrons are added to the other metal/ion system, the equilibrium shifts to remove the electrons.
A continuous flow of electrons occurs until either the metal or ions in the solution run out:
|
|
Zn(s) + Cu2+(aq) à Zn2+(aq) + Cu(s)
The half equations:
Zn2+(aq) +
2e-
Most (-)ve half cell - e's flow out of the half cell to the external wire - Equilibrium moves to LHS
Cu2+(aq) +
2e-
Most (+)ve half cell - e's flows into the half cell from the external wire - Equilibrium moves to RHS
Current flows in the opposite direction to the flow of electrons |
The salt bridge completes the circuit allowing ions to transfer between the half cells.
Usually made of filter paper soaked in KNO3
Non - metal / non - metal ion systems:
A half cell can be made from non metals and their ions:
2H+(aq) + 2e- ![]() H2(g)
H2(g)
| The hydrogen electrode: |
This is the reference electrode against which all other half cell electrode potential are measured (later)
For this reason the hydrogen electrode has a potential = 0
Conditions:
|
|
|
Other non - metal / non - metal ion half cells:
|
|
Fe2+(aq) +
e-
|
Qu 1 P185
Standard electrode potentials
These are measured against the reference electrode - The hydrogen half cell
By defining the hydrogen half cell = 0 all other half cells can be measured.
This measures its tendency to gain or lose electrons from its half cell with respect to hydrogen
Remember, the standard electrode potential is measured in volts, emf
Measuring standard electrode potentials:
|
|
The reading on the voltmeter gives the standard electrode potential of the Zn2+ / Zn half cell = - 0.76
Standard electrode potentials and cell reactions
Two half cells together give a cell. This gives a standard cell potential
The standard cell potential is the difference between the standard electrode potentials of the two half cells.
The cell reaction is the overall chemical reaction taking place
Example 1: A magnesium - copper cell is made by connecting 2 half cells together: Mg2+ / Mg and Cu2+ / Cu
1) Write out the half reactions. The more (-)ve value will produce electrons, the more positive will react with electrons.
|
Mg2+(aq) |
+ |
2e- |
|
Mg(s) |
Eq = -2.37 |
1 |
|
Cu2+(aq) |
+ |
2e- |
|
Cu(s) |
Eq = +0.34 |
2 |
2) Draw the direction of each of the half reactions:
|
|
| Mg2+(aq) | + | 2e- |
|
 |
Eq = -2.37 |
1 |
||
 |
+ | 2e- |
|
Cu(s) | Eq = +0.34 |
2 |
|
|
It is possible to write a reaction from this because this tells you that reaction 1 is going to move to the LHS and reaction 2 is going to move to the RHS.
3. Balanced chemical equation - Write out the 2 half reactions and the overall equation balancing with electrons
Write out the 2 half reactions in the direction they occur.
Mg(s) à Mg2+(aq) + 2e-
Cu2+(aq) + 2e- à Cu(s)
Balance the electrons (if necessary).
4) Add the half reactions
Add the reactions together and the electrons will cancel out:-
Mg(s) à Mg2+(aq) + 2e-
Cu2+(aq) + 2e- à Cu(s) add the half reactions
Mg(s) + Cu2+(aq) + 2e- à Mg2+(aq) + Cu(s) + 2e- electrons cancel out
Mg(s) + Cu2+(aq) à Mg2+(aq) + Cu(s)
5) Calculating emf of electrochemical cells:
Emf = Eqpos - Eqneg
Emf = + 0.34 - - 2.37
Emf = + 2.71v
Rules:
1) Write out the half reactions. The more (-)ve value will produce electrons, the more positive will react with electrons.
2) Draw the direction of each of the half reactions:
3) Writing balanced redox equation
4) Add the half reactions
5) Calculating emf: Eqcell = Eqpos - Eqneg
Example 2: A tin - iron cell is made by connecting 2 half cells together: Sn2+ / Sn and Fe3+ / Fe2+
1) Write out the half reactions. The more (-)ve value will produce electrons, the more positive will react with electrons.
| Sn2+(aq) | + | 2e- |
|
Sn2+(s) | Eq = -0.14 |
1 |
||
| Fe3+(aq) | + | e- |
|
Fe2+(aq) | Eq = +0.77 |
2 |
2) Draw the direction of each of the half reactions:
|
|
| Sn2+(aq) | + | 2e- |
|
 |
Eq = -0.14 |
1 |
||
 |
+ | e- |
|
Fe2+(aq) | Eq = +0.77 |
2 |
|
|
3. Balanced chemical equation - Write out the 2 half reactions and the overall equation balancing with electrons
Write out the 2 half reactions in the direction they occur.
Sn(s) à Sn2+(aq) + 2e-
Fe3+(aq) + e- à Fe2+(aq) x2
Balance the electrons.
4) Add the half reactions
Add the reactions together and the electrons will cancel out:-
Sn(s) à Sn2+(aq) + 2e-
2Fe3+(aq) + 2e- à 2Fe2+(aq)
Sn(s) + 2Fe3+(aq) + 2e- à Sn2+(aq) + 2Fe2+(aq) + 2e- electrons cancel out
Sn(s) + 2Fe3+(aq) à Sn2+(aq) + 2Fe2+(aq)
5) Calculating emf: Eqcell = Eqpos - Eqneg
Emf = Eqpos - Eqneg
Emf = + 0.77 - - 0.14
Emf = + 0.91v
Qu 1 P187
Electrochemical cells
These are used in every day life as a source of electricity, more commonly known as batteries.
They all work on the same principle - electrochemistry involving 2 redox reactions
The most basic cell is the copper / zinc redox system, commonly known as the Daniel cell:
|
|
The half equations:
Zn2+(aq) +
2e-
Cu2+(aq) +
2e-
Overall equation:
Zn(s) + Cu2+(aq) à Zn2+(aq) + Cu(s)
Emf = Eqpos - Eqneg Emf = + 0.34 - - 0.76 Emf = + 1.10v
|
Modern cells and batteries:
| 1) | Non - rechargable cells: | Provides electricity until the chemicals have reacted away. |
| 2) | Rechargable cells: | The chemicals react providing electricity until they have reacted away. The difference is that the chemicals can be regenerated by reversing the flow of electrons during charging. |
| 3) | Fuel cells: | The chemicals react providing electricity but the chemicals needed are constantly supplied. |
Fuel cells:
The hydrogen oxygen fuel cell:
 |
The half equations:
2H2O(l) +
2e-
1/2O2(g) + H2O(l)
+ 2e-
Overall equation:
H2(g) + 1/2O2(g) à H2O(l)
Emf = Eqpos - Eqneg Emf = + 0.40 - - 0.83 Emf = + 1.23v
|
Qu 1-2 P191
Development of fuel cell vehicles (FCV's):
CH3OH + H2O à 3H2 + CO2
Methanol as an alternative to hydrogen:
Advantages:
1) Liquids are easier to store than gases
2) Methanol can be produced from biomass
Problems:
1) Only generate a small amount of power
2) Produce CO2
Advantages of fuel cell vehicles:
1) Less CO2 produced
2) Normal hydrocarbons produce CO which needs to be removed by catalytic converters
3) Fuel cells are about 40 - 60% efficient / hydrocarbons are about 20%
Storage of hydrogen
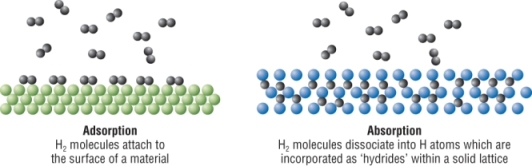
| Under pressure | Adsorbed onto a solid surface | Absorbed within a solid | |
| Hydrogen can be stored as a liquid under pressure. Very low temperatures are required to keep them as liquids. A large 'thermos flask' would be needed to prevent it from boiling. | Like a catalyst holds molecules in place. This means that the hydrogen molecules occupy a smaller volume then as a gas. | Similar to before, the hydrogen molecules absorb into the material meaning that the hydrogen occupies a smaller volume. | |
 |
|||
Limitations of hydrogen fuel cells:
The hydrogen economy:
Qu 1 - 2 P193 / Qu 1 - 7 P195 / Qu 1 - 6 P196 - 197