
=
Introduction:
In AS we covered the characteristics of an equilibrium:
In AS we discovered that Le Chatelier’s Principle was given as a way of determining how changes in the conditions can affect the extent to which a reaction will go.
|
|
Kc |
= |
[PRODUCTS]p | |
| [REACTANTS]r |
| aA | + | bB | D | cC | + | dD |
| Kc |
= |
[C]c [D]d |
| [A]a [B]b |
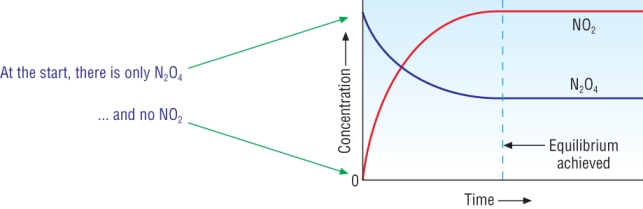
Approaching equilibrium
| N2O4(g) | D | 2NO2(g) |
| Initially: |
|
|
| As the reaction proceeds: |
|
|
| At equilibrium: |
|
Writing expressions for Kc:
| N2O4(g) | D | 2NO2(g) |
| Kc |
= |
[NO2]2 |
| [N2O4] |
| 2SO2(g) | + | O2(g) | D | 2SO3(g) |
| Kc |
= |
[SO3]2 |
| [SO2]2 [O2] |
| H2(g) | + | I2(g) | D | 2HI(g) |
| Kc |
= |
[HI]2 |
| [H2] [I2] |
Units of Kc
| Kc | = | [NO2]2 | Kc | = | [SO3]2 | Kc | = | [HI]2 | ||||||
| [N2O4]1 | [SO2]2 [O2] | [H2] [I2] | ||||||||||||
| Kc | = | mol dm-3 x mol dm-3 | Kc | = | (mol dm-3)2 | Kc | = | mol dm-3 mol dm-3 | ||||||
| mol dm-3 | (mol dm-3)2 | mol dm-3 | mol dm-3 | mol dm-3 | ||||||||||
| Kc | = |
mol dm-3 x |
Kc | = |
|
Kc | = |
|
||||||
|
|
|
mol dm-3 |
|
|
||||||||||
| Kc | = | mol dm-3 | Kc | = | 1 | Kc | = | No units | ||||||
| mol dm-3 | ||||||||||||||
| Kc | = | dm3 mol-1 | ||||||||||||
Qu 1 P127
Determining Kc from equilibrium concentrations:
1) Hydrogen, Iodine, Hydrogen iodide equilibrium:
| Reaction: | H2(g) | + | I2(g) | D | 2HI(g) |
| Equilibrium concentrations: | 0.140 | 0.040 | 0.320 |
| Kc | = | [HI]2 | Kc | = | [HI]2 | |||||
| [H2] [I2] | [H2] [I2] | |||||||||
| Kc | = | (0.320)2 | Kc | = | mol dm-3 mol dm-3 | |||||
| 0.140 x 0.040 | mol dm-3 | mol dm-3 | ||||||||
| Kc | = | 18.3 | Kc | = |
|
|||||
|
|
|
|||||||||
| Kc | = | No units | ||||||||
2) N2O4 / NO2 equilibrium: Given the number of moles at equilibrium in a volume of 2dm3.
| Reaction: | N2O4(g) | D | 2NO2(g) |
| Mole quantities at equilibrium: | 0.400 | 3.20 | |
| Equilibrium concentrations: | 0.400 / 2 | 3.20 / 2 | |
| Equilibrium concentrations: | 0.20 | 1.60 |
| Kc | = | [NO2]2 | Kc | = | [NO2]2 | |||||
| [N2O4] | [N2O4] | |||||||||
| Kc | = | (1.60)2 | Kc | = | mol dm-3 mol dm-3 | |||||
| 0.20 | mol dm-3 | |||||||||
| Kc | = | 12.8 | mol dm-3 | Kc | = |
|
||||
|
|
||||||||||
| Kc | = | mol dm-3 | ||||||||
Calculating the quantities and concentrations present at equilibrium:
3) Hydrogen, Iodine, Hydrogen iodide equilibrium:
| Reaction: | H2(g) | + | I2(g) | D | 2HI(g) |
| At start: | 0.60 | 0.40 | 0.0 | ||
| At equilibrium: | 0.28 | 0.08 | 0.64 | ||
| Reacted: | 0.32 | 0.32 | 0.64 | ||
| Equilibrium concentrations: | 0.28 | 0.08 | 0.64 |
| Kc | = | [HI]2 | Kc | = | [HI]2 | |||||
| [H2] [I2] | [H2] [I2] | |||||||||
| Kc | = | (0.64)2 | Kc | = | mol dm-3 mol dm-3 | |||||
| 0.28 x 0.08 | mol dm-3 | mol dm-3 | ||||||||
| Kc | = | 18.3 | Kc | = |
|
|||||
|
|
|
|||||||||
| Kc | = | No units | ||||||||
Qu 1 - 2 P129
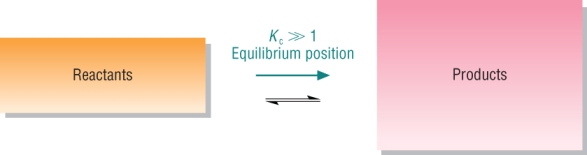
The equilibrium position and Kc
What is the significance of a Kc value?
|
|
Products favoured: Kc > 1
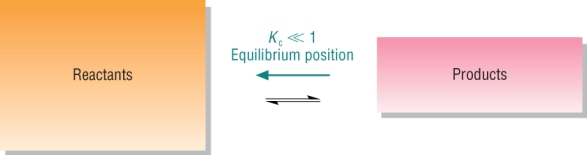
Reactants favoured: Kc < 1
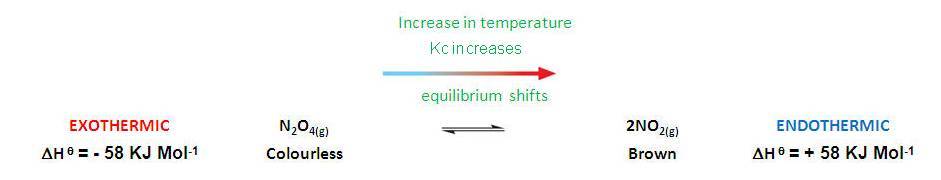
How do changes in temperature affect Kc?
1) Endothermic reactions:
|
|
Kc increases and vice versa |
2) Exothermic reactions:
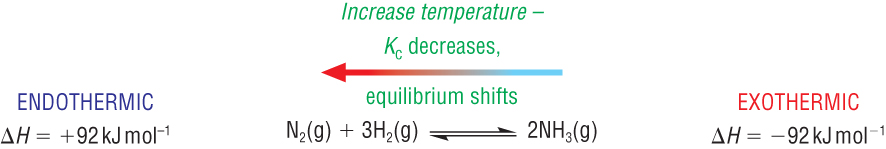
|
|
Kc decreases and vice versa |
Qu 1 P131
The equilibrium constant, Kc, and the rate constant, k
How does a change in concentration and pressure affect Kc?
Kc is unaltered by changes in concentration and pressure
1) Changes in concentration:
A change in Concentration has no effect on the equilibrium constant.
Remember if a reactant / product is added the equilibrium shifts to the opposite direction to keep the 'proportions' the same - Kc is unchanged:
| Reaction: | N2O4(g) | D | 2NO2(g) |
| [NO2] = 1.60 Mol dm-3 | [NO2] = 1.60 Mol dm-3 | ||||||||
| [N2O4] = 0.20 Mol dm-3 | Double [ ] : | [N2O4] = 0.40 Mol dm-3 | |||||||
| Kc | = | [NO2]2 | Kc | = | [NO2]2 | ||||
| [N2O4] | [N2O4] | ||||||||
| Kc | = | (1.60)2 | Kc | = | (3.20)2 | ||||
| 0.20 | 0.40 | ||||||||
| Kc | = | 12.8 |
Mol dm-3 |
Kc | = | 6.4 | Mol dm-3 | ||
If the concentrations are doubled, the system is no longer at equilibrium, Kc = 12.8
To bring Kc back from 6.4 to 12.8:
The system must increase [NO2] and decrease [N2O4]
Remember if a reactant / product is added the equilibrium shifts to the opposite direction to keep the 'proportions' the same - Kc is unchanged:
2) Changes in Pressure:
A change in Pressure has no effect on the equilibrium constant.
If pressure is doubled - the volume is halved - meaning that the concentrations will have doubled
| Reaction: | N2O4(g) | D | 2NO2(g) |
| [NO2] = 1.60 Mol dm-3 | Double [ ] : | [NO2] = 3.20 Mol dm-3 | |||||||
| [N2O4] = 0.20 Mol dm-3 | [N2O4] = 0.40 Mol dm-3 | ||||||||
| Kc | = | [NO2]2 | Kc | = | [NO2]2 | ||||
| [N2O4] | [N2O4] | ||||||||
| Kc | = | (1.60)2 | Kc | = | (3.20)2 | ||||
| 0.20 | 0.40 | ||||||||
| Kc | = | 12.8 |
Mol dm-3 |
Kc | = | 25.6 | Mol dm-3 | ||
If the pressure is doubled, the system is no longer at equilibrium, Kc = 12.8
To bring Kc back from 25.6 to 12.8:
The system must increase [NO2] and decrease [N2O4]
Remember if a reactant / product is added the equilibrium shifts to the opposite direction to keep the 'proportions' the same - Kc is unchanged:
How does the presence of a catalyst affect Kc?
A catalyst has no effect on the equilibrium constant.
A catalyst speeds up both the forward and reverse reaction.
Equilibrium is achieved more quickly.
The equilibrium constant Kc, and the rate constant, k
These 2 constants tell us the most important things in the chemical industry:
| a) | Equilibrium | How far |
| b) | Rates | How fast |
a) The equilibrium constant, Kc
Kc indicates the position of the equilibrium:
| Large Kc | Products predominate | |
| Small Kc | Reactants predominate |
Remember LCP:
| Endothermic | Kc increases with an increase in temperature (increases products) | |
| Exothermic | Kc decreases with an increase in temperature (decreases products) |
Kc can be written from the balanced chemical equation
b) The rate constant, k
k is a measure of the rate of a reaction:
| Large k | Fast rate | |
| Small k | Slow rate |
Remember:
| k increases with an increase in temperature - Rate increases with an increase in temperature | |
| k decreases with a decrease in temperature - Rate decreases with a decrease in temperature |
k can only be determined experimentally from the rate equation
The importance of compromise:
The 2 desirable outcomes are a) increasing rate and b) increasing the amount:
a) Increasing the rate:
| Increasing temperature: Increases the rate of production of product - desirable |
|
|
|
|
|
|
| Increasing temperature: Decreases the amount of product made - undesirable |
b) Increasing the amount:
| Decreasing the temperature: Increases the amount of product made - desirable |
|
|
|
|
|
|
| Decreasing temperature: Decreases the rate of production of product - undesirable |
The compromise is:
| Moderate temperature: | k increases moderately - Rate increases by a moderate amount |
| Moderate temperature: | Kc decreases by a moderate amount - allowing a moderate yield |
Qu 1 P133 / Qu 4 - 5 P159 / Qu 3 - 4 P161